NCERT Solutions for Class 11 Physics Chapter 13 Oscillations has been published by Aglasem. You can now download the Class 11 Physics Ch 13 Questions and Answers PDF here. This NCERT Solutions for Class 11 Physics contains answers of all questions asked in Chapter 13 in textbook, Physics Part II. Therefore you can refer it to solve Oscillations exercise questions and learn more about the topic.
NCERT Solutions for Class 11 Physics Chapter 13 Oscillations
Class – Class 11
Subject – Physics
Chapter – Ch 13
Chapter Name – Oscillations
Book – Physics Part II
Study Material – NCERT Solutions



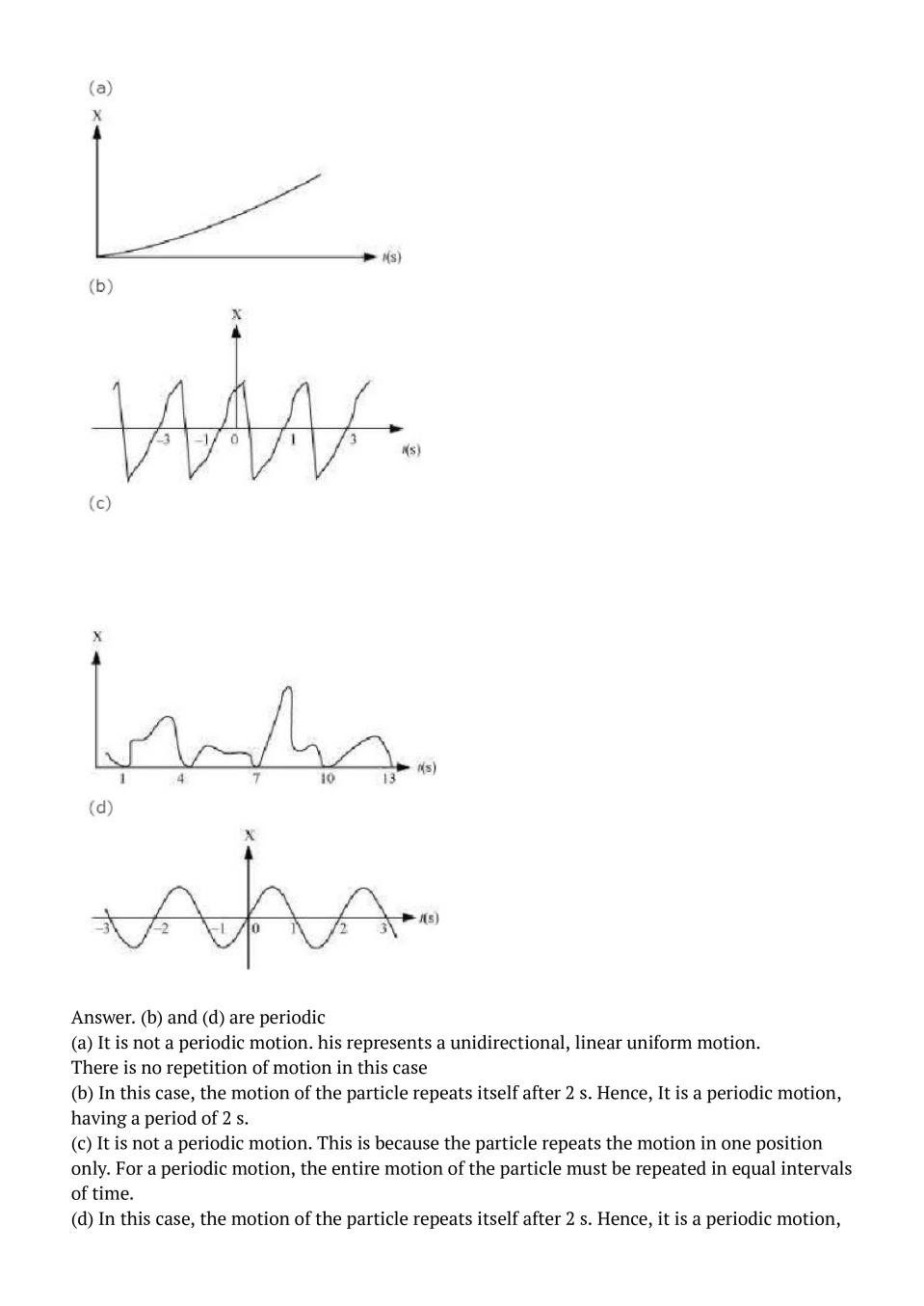






















NCERT Solutions for Class 11 Physics Chapter 13 PDF
While you can read NCERT Solutions for Class 11 Physics Ch 13 for all exercises here on aglasem. You can also download this NCERT Solutions PDF to refer ncer question answer at anytime when you study Oscillations. Here it is.
NCERT Solutions for Class 11 Physics Chapter 13 PDF Download Link – Click Here to Download Solutions PDF
How to download NCERT Solutions for Class 11 Physics Chapter 13 PDF?
You can download the complete NCERT solutions for chapter 13 of this NCERT Book i.e. Physics Part II with following steps.
- First search NCERT Solutions for Class 11 Physics Ch 13 PDF aglasem and come to this page.
- Now you will see the exercise questions answers of Oscillations and download pdf link on it.
- Click the Download PDF link to obtain the Oscillations questions with answers document.
NCERT Solutions for Class 11 Physics
There are more chapters to study besides Oscillations in this subject. So here are NCERT solutions for all topics of Physics taught in 11th class here at aglasem.
- Chapter 1 Units and Measurements
- Chapter 2 Motion in a Straight Line
- Chapter 3 Motion in a Plane
- Chapter 4 Laws of Motion
- Chapter 5 Work, Energy and Power
- Chapter 6 System of Particles and Rotational Motion
- Chapter 7 Gravitation
- Chapter 8 Mechanical Properties of Solids
- Chapter 9 Mechanical Properties of Fluids
- Chapter 10 Thermal Properties of Matter
- Chapter 11 Thermodynamics
- Chapter 12 Kinetic Theory
- Chapter 13 Oscillations
- Chapter 14 Waves
NCERT Solutions for Class 11
Just like you got Physics Ch 13 solutions here. You can see exercise questions answers of other subjects and their topics too on aglasem. Here are NCERT solutions for all subjects of 11th standard NCERT books.
- NCERT Solutions for Class 11 Accountancy
- NCERT Solutions for Class 11 Biology
- NCERT Solutions for Class 11 Business Studies
- NCERT Solutions for Class 11 Chemistry
- NCERT Solutions for Class 11 Economics
- NCERT Solutions for Class 11 English
- NCERT Solutions for Class 11 Geography
- NCERT Solutions for Class 11 Hindi
- NCERT Solutions for Class 11 History
- NCERT Solutions for Class 11 Maths
- NCERT Solutions for Class 11 Physics
- NCERT Solutions for Class 11 Political Science
- NCERT Solutions for Class 11 Psychology
- NCERT Solutions for Class 11 Sociology
NCERT Solutions for Class 11 Physics Chapter 13 – An Overview
The key highlights of this study material are as follows.
| Aspects | Details |
|---|---|
| Class | Class 11 |
| Subject | Physics |
| Chapter Number | Ch 13 |
| Chapter Name | Oscillations |
| Book Name | Physics Part II |
| Book By | NCERT (National Council of Educational Research and Training) |
| Educational Resource Here | NCERT Solutions of Class 11 Physics Ch 13 for All Exercise |
| More Questions Answers of This Subject | NCERT Solutions for Class 11 Physics |
| Download Book Chapter PDF | NCERT Book Class 11 Physics Chapter 13 |
| All Questions Answers For This Class | NCERT Solutions for Class 11 |
| Complete Solutions | NCERT Solutions |
If you have any queries on NCERT Solutions for Class 11 Physics Chapter 13 Oscillations, then please ask in comments below.
To get study material, exam alerts and news, join our Whatsapp Channel.


