Class 12 Chemistry Coordination Compounds – Get here the Notes, Question & Practice Paper of Class 12 Chemistry for topic Coordination Compounds Notes. Coordination Compounds Notes for Class 12 Chemistry are here. You can download the Coordination Compounds Notes PDF to study all the topics in this chapter. Moreover the class 12 Chemistry notes include chapter summary, definitions, examples, and key pointers for Coordination Compounds. Thus if you are studying class Chemistry (रसायन शास्त्र), then the Coordination Compounds notes will help you easily understand the topic and ace it.
Class 12 Chemistry Notes for Coordination Compounds
Coordination Compounds is a critical part in the study of Chemistry. In India, it is taught in class. Therefore the class 12 Notes for Chemistry topic Coordination Compounds have been compiled by teachers and field experts. They explain the complete chapter of Coordination Compounds in one-shot. Whether you are studying the topic Coordination Compounds to complete your class syllabus, or for any competitive exam like JEE, NEET, UPSC, you can simply refer these notes to complete the chapter in one-shot!
Coordination Compounds Notes Download Link – Click Here to Download PDF
Coordination Compounds Notes for Class 12 Chemistry PDF
The PDF of Coordination Compounds class 12 notes is as follows. You can view the document here and also download it to use it anytime for future reference whenever you want to brush up your concepts of Chemistry.
Class 12 Chemistry Notes for Coordination Compounds View Download




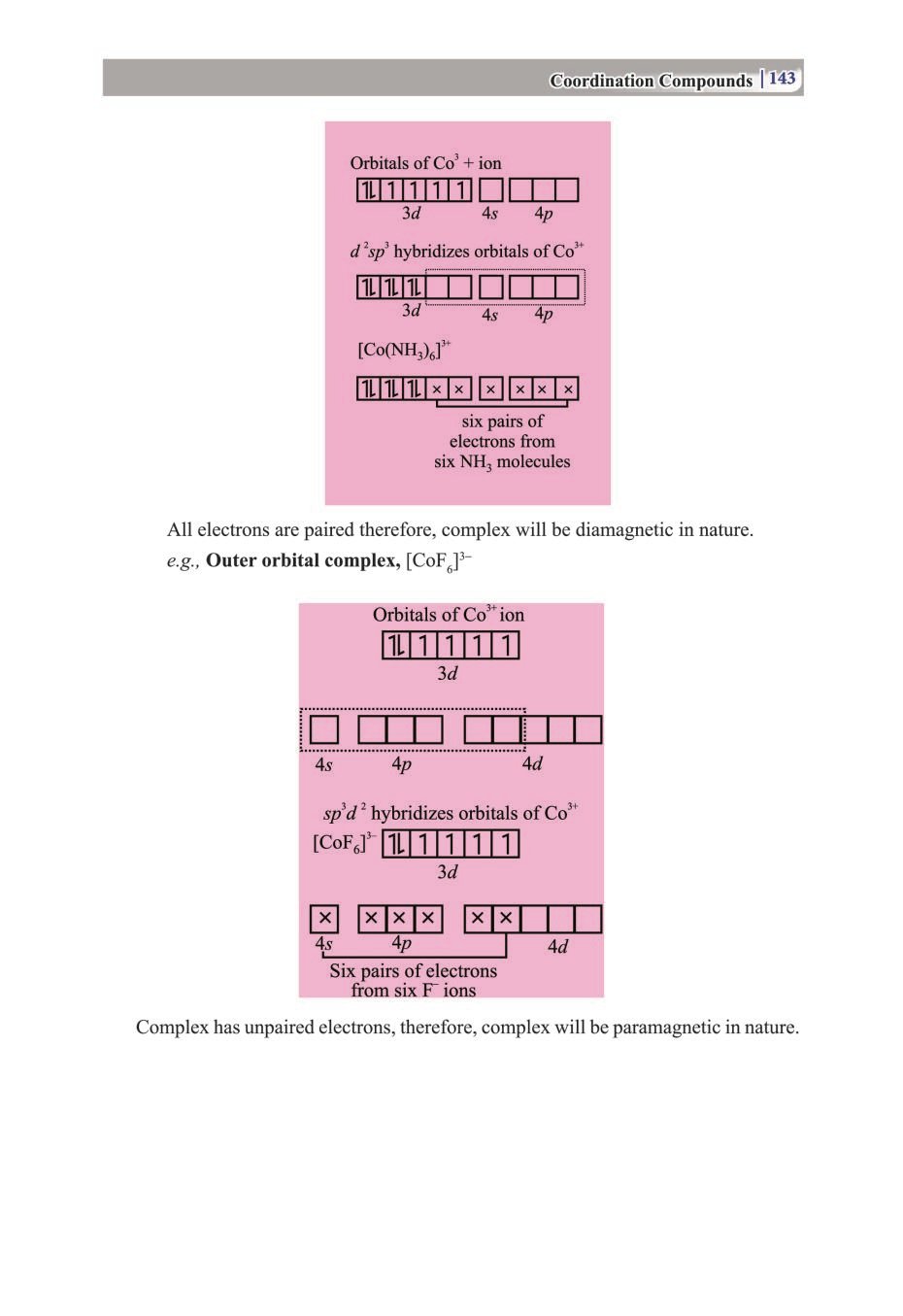
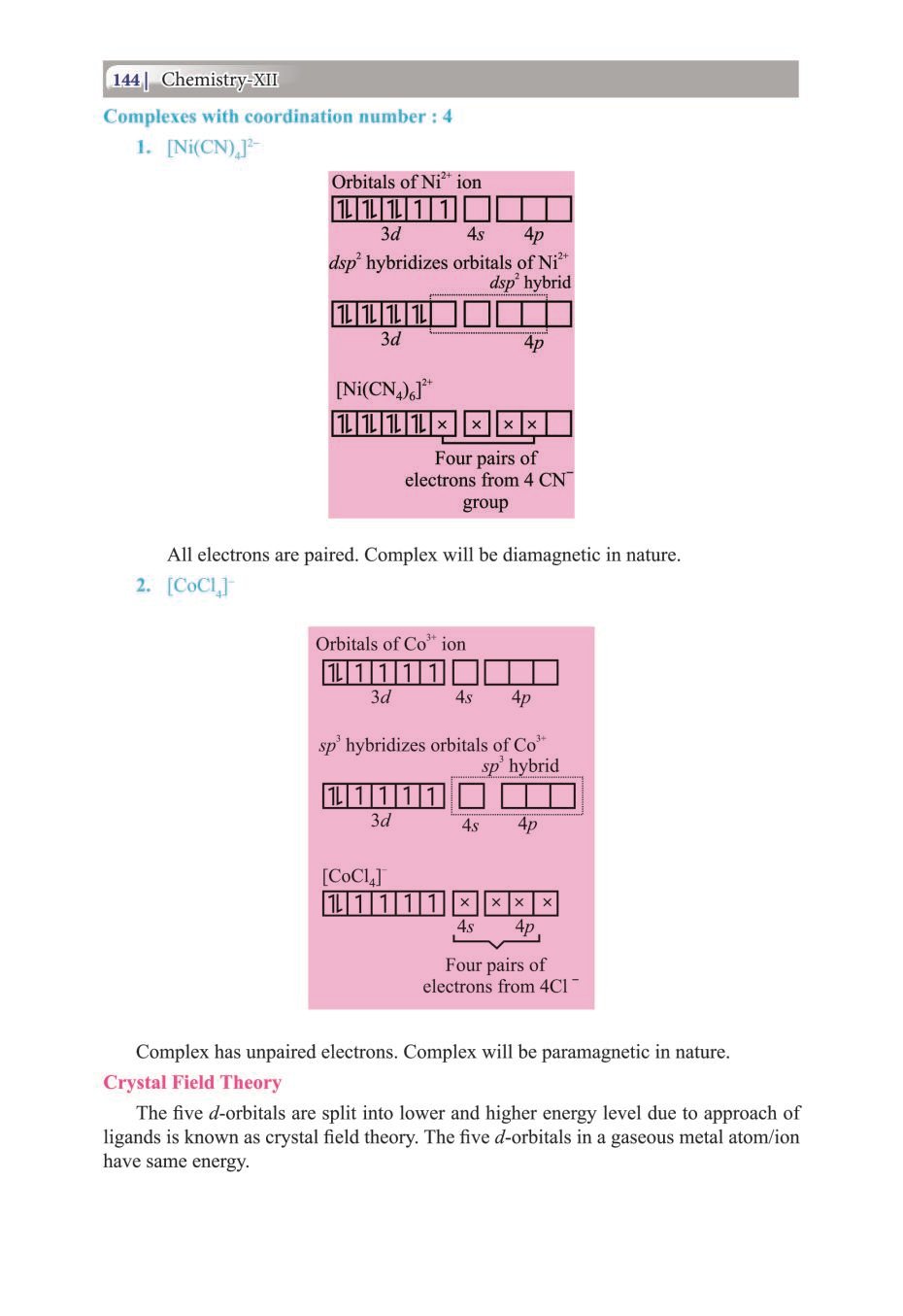









Candidates who are ambitious to qualify the Class 12 with good score can check this article for Notes, Study Material, Practice Paper. Above we provided the link to access the Notes, Important Question and Practice Paper of Class 12 Chemistry for topic Coordination Compounds.
All Topics Class 12 Chemistry Notes
Chapter wise notes for Chemistry (रसायन शास्त्र) are given below.
- Alcohol Phenol and Ethers
- Aldehydes Ketones And Carboxylic Acids
- Amines
- Biomolecules
- Chemical Kinetics
- Chemistry in Everyday Life
- Coordination Compounds
- d- and f-Block Elements
- Electrochemistry Concepts
- General Principles and Processes of Isolation of Elements
- Haloalkanes and Haloarenes
- p-Block Elements
- Polymers
- Solutions
- Surface Chemistry
- The Solid State
Class 12 Notes for All Subjects
- Class 12 Biology Notes
- Class 12 Business Studies Notes
- Class 12 Chemistry Notes
- Class 12 Economics Notes
- Class 12 English Notes
- Class 12 Geography Notes
- Class 12 Maths Notes
- Class 12 Physics Notes
- Class 12 Sociology Notes
NCERT Solutions for Class 12 Chemistry Coordination Compounds
The Coordination Compounds notes here help you solve the questions and answers. Also, you can complete the class 12 Coordination Compounds worksheet using the same. In addition you will also tackle CBSE Class 12 Chemistry Important Questions with these class 12 notes.
However if you still need help, then you can use the NCERT Solutions for Class 12 Chemistry Coordination Compounds to get all the answers. Coordination Compounds solutions contain questions, answers, and steps to solve all questions.
Notes for All Classes
Coordination Compounds Notes for Class 12 Chemistry – An Overview
| Name of Topic | Coordination Compounds |
| Class | 12 |
| Subject | Chemistry |
| All Class 12 Chemistry Notes | Class 12 Chemistry Notes |
| All Class 12 Notes | Class 12 Notes |
Class 12 Coordination Compounds Notes for All Boards
You can use the class 12 Chemistry notes of Coordination Compounds for all boards.
The education boards in India for which Coordination Compounds notes are relevant are – CBSE, CISCE, AHSEC, CHSE Odisha, CGBSE, HBSE, HPBOSE, PUE Karnataka, MSBSHSE, PSEB, RBSE, TBSE, UPMSP, UBSE, BIEAP, BSEB, GBSHSE, GSEB, JAC, JKBOSE, KBPE, MBOSE, MBSE, MPBSE, NBSE, DGE TN, TSBIE, COHSEM, WBCHSE.
Therefore you can refer to these notes as CBSE, CISCE, AHSEC, CHSE Odisha, CGBSE, HBSE, HPBOSE, PUE Karnataka, MSBSHSE, PSEB, RBSE, TBSE, UPMSP, UBSE, BIEAP, BSEB, GBSHSE, GSEB, JAC, JKBOSE, KBPE, MBOSE, MBSE, MPBSE, NBSE, DGE TN, TSBIE, COHSEM, WBCHSE notes for class Class 12 / Class / Chemistry for the topic Coordination Compounds.
To get study material, exam alerts and news, join our Whatsapp Channel.

