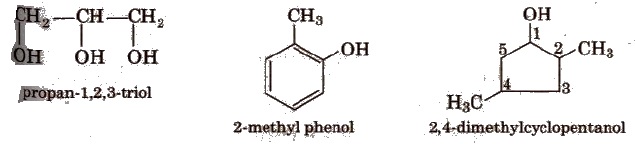
Alcohols and Phenols
Alcohols and phenols are formed when a hydrogen atom in hydrocarbon, aliphatic and aromatic respectively, is replaced by hydroxyl group (-OR group).
Classification of Alcohols and Phenols
In alcohols, -OR group is attached to Sp3 hybridised carbon. These alcohols are usually classified as primary, secondary and tertiary alcohols.
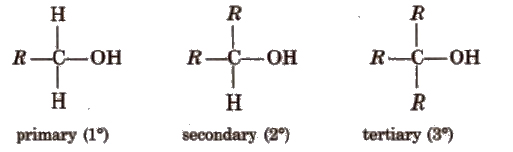 Alcohols may be
(i) monohydric-containing one – OR group,
(ii) dihydric-containing two – OR groups and
(iii) polyhydric-containing three or more -OR groups.
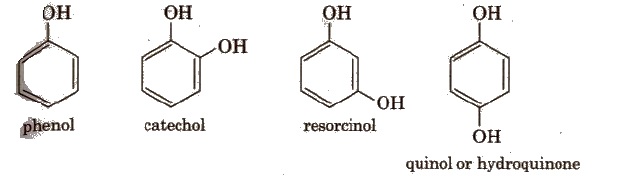
In phenols, -OR group is attached to Sp2 hybridised carbon. These may also be monohydric, dihydric, etc. The dihydric phenol further rosy be ortho, meta’ or para derivative.
Alcohols may be
(i) monohydric-containing one – OR group,
(ii) dihydric-containing two – OR groups and
(iii) polyhydric-containing three or more -OR groups.
In phenols, -OR group is attached to Sp2 hybridised carbon. These may also be monohydric, dihydric, etc. The dihydric phenol further rosy be ortho, meta’ or para derivative.
 In allylic alcohols, – OH group is attached to sp3 hybridised carbon but next to C=C bond.
e.g., CH2 = CH – CH2OH, Benzylic alcoho1(C6H5CH2OH)
Structure of Alcohols and Phenols
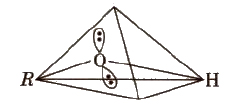
The oxygen atom of alcohols is Sp3 hybridised and they have tetrahedral position of hybrid atomic orbitals .
In allylic alcohols, – OH group is attached to sp3 hybridised carbon but next to C=C bond.
e.g., CH2 = CH – CH2OH, Benzylic alcoho1(C6H5CH2OH)
Structure of Alcohols and Phenols
The oxygen atom of alcohols is Sp3 hybridised and they have tetrahedral position of hybrid atomic orbitals .
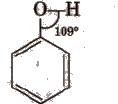
 The value of LROH bond angle depends upon the R group. For methyl alcohol, it is (∠C – O – H) 108.9° due to repulsion of lone pairs.
In phenols, the – OH group is attached to Sp2 hybridised carbon and thus, the C – O bond acquires a partial double bond character.
The value of LROH bond angle depends upon the R group. For methyl alcohol, it is (∠C – O – H) 108.9° due to repulsion of lone pairs.
In phenols, the – OH group is attached to Sp2 hybridised carbon and thus, the C – O bond acquires a partial double bond character.
 Nomenclature of Alcohols and Phenol
In IUPAC, system, alcohol or alkanols are named by replacing the last word ‘e’ of the corresponding alkane by ‘ol’. e.g.,
Nomenclature of Alcohols and Phenol
In IUPAC, system, alcohol or alkanols are named by replacing the last word ‘e’ of the corresponding alkane by ‘ol’. e.g.,
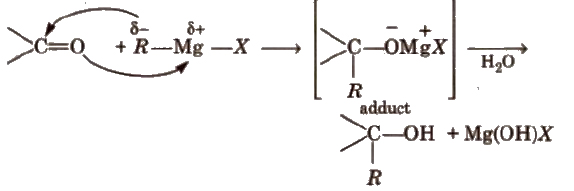
 Preparation of Alcohols
(i) From alkenes
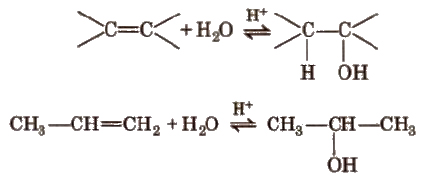
(a) By acid catalysed hydration in accordance with Markownikoff’s rule.
Preparation of Alcohols
(i) From alkenes
(a) By acid catalysed hydration in accordance with Markownikoff’s rule.
 Mechanism
Step I Protonation of alkene by attack of H3O+
Mechanism
Step I Protonation of alkene by attack of H3O+
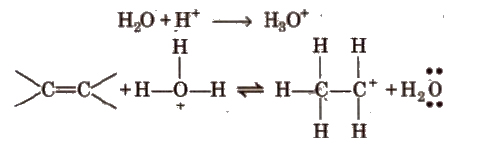 Step II Nucleophilic attack
Step II Nucleophilic attack
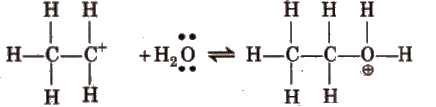 Step III Deprotonation to form an alcohol
Step III Deprotonation to form an alcohol
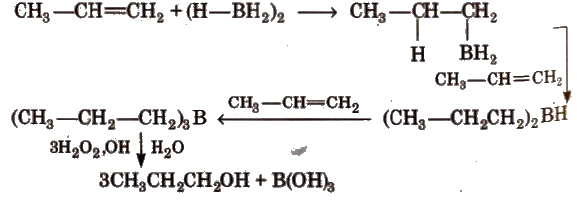
 (b) By hydroboration-oxidation
(b) By hydroboration-oxidation
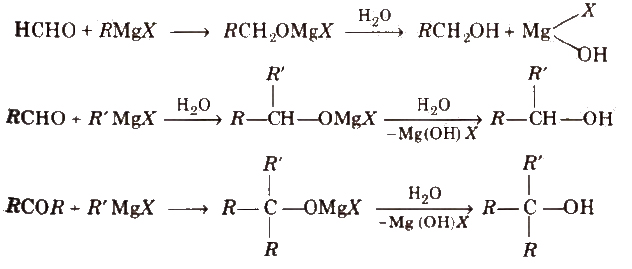
 (ii) From carbonyl compounds
(a) By reduction of aldehydes and ketones
(ii) From carbonyl compounds
(a) By reduction of aldehydes and ketones
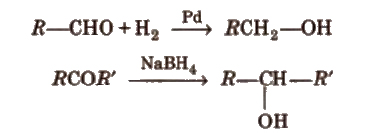 Aldehydes yield primary alcohols whereas ketones give secondary alcohols, when subjected to reduction.
(b) By reduction of carboxylic acids and ester
Aldehydes yield primary alcohols whereas ketones give secondary alcohols, when subjected to reduction.
(b) By reduction of carboxylic acids and ester
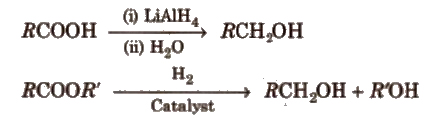 Reduction of aldehyde, ketones and esters with No Alcohol is called Bouveault-blanc reduction.
Reduction of aldehyde, ketones and esters with No Alcohol is called Bouveault-blanc reduction.
 The reaction produces a primary alcohol with methanol, a secondary alcohol with aldehydes (except methanal) and tertiary alcohol with ketones
The reaction produces a primary alcohol with methanol, a secondary alcohol with aldehydes (except methanal) and tertiary alcohol with ketones
 (iv) Hydrolysis of alkyl halides
R – X + KOH(aq) → ROH + KX
To avoid dehydrohalogenation of RX, mild alkalies like moist
Ease of hydrolysis of alkyl halides RI > R – Br > RCI > and t > s > p alkyl halides.
(v) Hydrolysis of ethers
(iv) Hydrolysis of alkyl halides
R – X + KOH(aq) → ROH + KX
To avoid dehydrohalogenation of RX, mild alkalies like moist
Ease of hydrolysis of alkyl halides RI > R – Br > RCI > and t > s > p alkyl halides.
(v) Hydrolysis of ethers
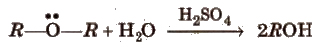 (vi) From primary amines By treatment with nitrous acid.
(vi) From primary amines By treatment with nitrous acid.
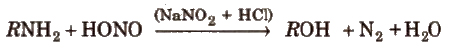 Methylamine does not give methyl alcohol when treated with HNO2. It gives CH3OCH3 and CH3ONO.
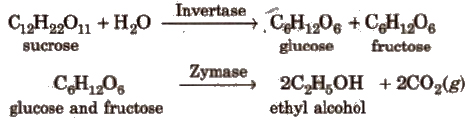
(vii) By alcoholic fermentation
Methylamine does not give methyl alcohol when treated with HNO2. It gives CH3OCH3 and CH3ONO.
(vii) By alcoholic fermentation
 Preparation of Phenols
(i) From haloarenes
Preparation of Phenols
(i) From haloarenes
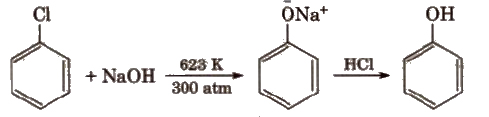 (ii) From benzene sulphonic acid
(ii) From benzene sulphonic acid
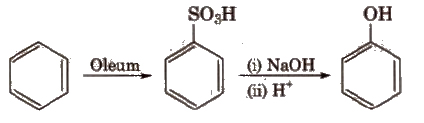 (iii) From diazonium salts
(iii) From diazonium salts
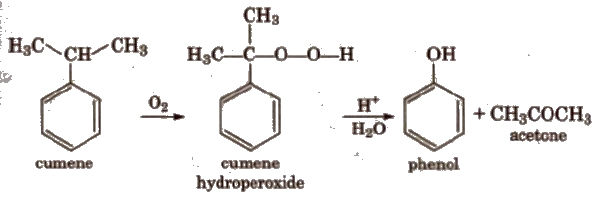
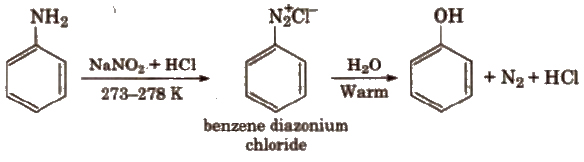 (iv) From cumene
(iv) From cumene
 Physical Properties of Alcohols
1. Lower alcohols are colourless liquids, members from C5 – C11 are oily liquids and higher members are waxy solids.
2. The hydroxyl groups in alcohols can form H-bonds with water, so alcohols are miscible with water. The solubility decreases with increase in molecular mass.
Physical Properties of Alcohols
1. Lower alcohols are colourless liquids, members from C5 – C11 are oily liquids and higher members are waxy solids.
2. The hydroxyl groups in alcohols can form H-bonds with water, so alcohols are miscible with water. The solubility decreases with increase in molecular mass.
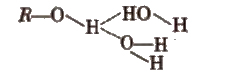 3. Boiling points of alkanes are higher than expected because of the presence of intermolecular hydrogen bonding in the polar molecules.
[The boiling point decreases in the order 1° > 2° > 3° as the van der Waals’ forces of attraction decreases]
Physical Properties of Phenols
1. These are colourless liquids or crystalline solids but become coloured due to slow oxidation with air.
2. Phenol is also called carbolic acid.
3. Because of the presence of polar -OH bond, phenols form intermolecular H-bonding with other phenol molecules and with water.
Chemical Reactions of Alcohols and Phenols
(i) Reactions involving cleavage of O – H Bond
(a) Acidity of alcohols and phenols
3. Boiling points of alkanes are higher than expected because of the presence of intermolecular hydrogen bonding in the polar molecules.
[The boiling point decreases in the order 1° > 2° > 3° as the van der Waals’ forces of attraction decreases]
Physical Properties of Phenols
1. These are colourless liquids or crystalline solids but become coloured due to slow oxidation with air.
2. Phenol is also called carbolic acid.
3. Because of the presence of polar -OH bond, phenols form intermolecular H-bonding with other phenol molecules and with water.
Chemical Reactions of Alcohols and Phenols
(i) Reactions involving cleavage of O – H Bond
(a) Acidity of alcohols and phenols
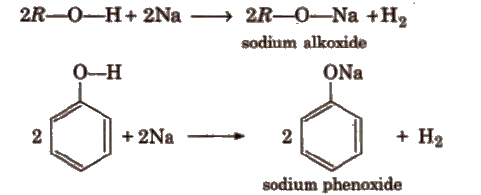 Alcohols are weaker acids than water due to +1 group present in alcohols, which decreases the polarity of -O-H bond.
Acid strength of alcohols
Alcohols are weaker acids than water due to +1 group present in alcohols, which decreases the polarity of -O-H bond.
Acid strength of alcohols
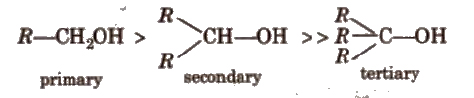 Electron releasing group increases electron density on oxygen to decrease the polarity of – OH bond.
Order of acidity is
RCOOH > H2CO3 > C6H5OH > H2O > R – OH.
Phenol is more acidic than alcohols due to stabilisation of phenoxide ion through resonance. Presence of electron withdrawing group increases the acidity of phenol by ,
stabilising phenoxide ion while presence of electron releasing group decreases the acidity of phenol by destabilising phenoxide ion.
Thus. increasing acidic strength is
o-cresol < p-cresol < m-cresol < phenol < o-nitrophenol < 2, 4. 6.trinitrophenol (picric acid)
Higher Ka and lower pKa value corresponds to the stronger acid.
(b) Esterification
Electron releasing group increases electron density on oxygen to decrease the polarity of – OH bond.
Order of acidity is
RCOOH > H2CO3 > C6H5OH > H2O > R – OH.
Phenol is more acidic than alcohols due to stabilisation of phenoxide ion through resonance. Presence of electron withdrawing group increases the acidity of phenol by ,
stabilising phenoxide ion while presence of electron releasing group decreases the acidity of phenol by destabilising phenoxide ion.
Thus. increasing acidic strength is
o-cresol < p-cresol < m-cresol < phenol < o-nitrophenol < 2, 4. 6.trinitrophenol (picric acid)
Higher Ka and lower pKa value corresponds to the stronger acid.
(b) Esterification
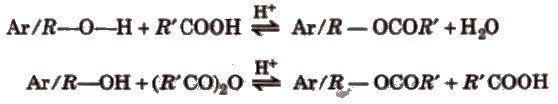
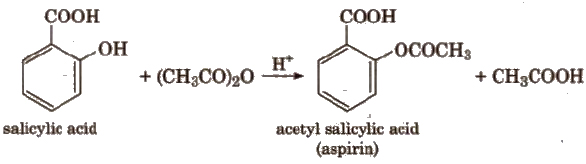
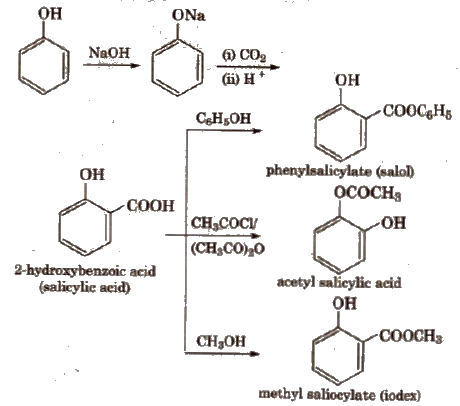
 The reaction with R’COOH and (R’ CO)2O is reversible, so cone, H2SO4 is used to remove water.
The reaction with R’ COCI is carried out in the presence of pyridine so as to neutralise HCI which is formed during the reaction.
The introduction of acetyl (CH3CO-) group in phenols is known as acetylation.
Acetylation of salicylic acid produces aspirin.
The reaction with R’COOH and (R’ CO)2O is reversible, so cone, H2SO4 is used to remove water.
The reaction with R’ COCI is carried out in the presence of pyridine so as to neutralise HCI which is formed during the reaction.
The introduction of acetyl (CH3CO-) group in phenols is known as acetylation.
Acetylation of salicylic acid produces aspirin.
 (ii) Reaction involving cleavage of C-O bond in alcohols In these reactions, the reactivity order of different alcohols :
(ii) Reaction involving cleavage of C-O bond in alcohols In these reactions, the reactivity order of different alcohols :
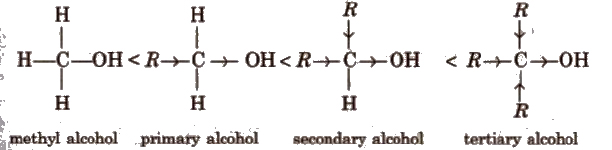 Alkyl group due to +1 effect increases the electron density on the carbon and oxygen atom of C-OH bond. As a result, the bond cleavage becomes easy. Greater the number of alkyl groups present, more will be the reactivity of alcohol. Thus, the relative order of reactivity of the alcohols is justified.
(a) Reaction with halogen acids Alcohols can be converted into haloalkanes by the action of halogen acids.
R – OH + HX (HCI, HBr, HI) → R-X +H2O
For a given alcohol order of reactivity of HX is
H-1 > H-Br > H-Cl
For a given halogen acid order of reactivity of alcohols
Tertiary > Secondary > Primary
Lucas test
Alkyl group due to +1 effect increases the electron density on the carbon and oxygen atom of C-OH bond. As a result, the bond cleavage becomes easy. Greater the number of alkyl groups present, more will be the reactivity of alcohol. Thus, the relative order of reactivity of the alcohols is justified.
(a) Reaction with halogen acids Alcohols can be converted into haloalkanes by the action of halogen acids.
R – OH + HX (HCI, HBr, HI) → R-X +H2O
For a given alcohol order of reactivity of HX is
H-1 > H-Br > H-Cl
For a given halogen acid order of reactivity of alcohols
Tertiary > Secondary > Primary
Lucas test
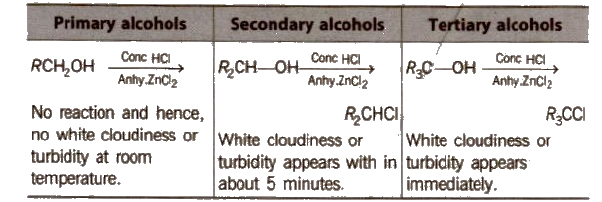 (b) Reaction with phosphorus halides
(b) Reaction with phosphorus halides
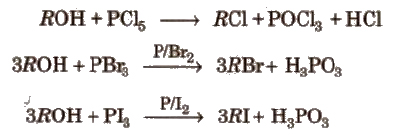 (c) Reaction with thionyl chloride
(c) Reaction with thionyl chloride
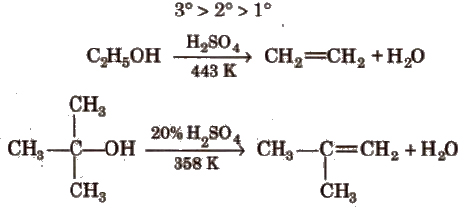
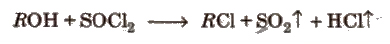 d) Dehydration of alcohols It requires acid catalyst and the reaction proceeds via intermediate carbonium ion. Acidic catalyst converts hydroxyl group into a good leaving group.
Since, the rate determining step is the formation of carbocation, the ease of dehydration is
d) Dehydration of alcohols It requires acid catalyst and the reaction proceeds via intermediate carbonium ion. Acidic catalyst converts hydroxyl group into a good leaving group.
Since, the rate determining step is the formation of carbocation, the ease of dehydration is
 Mechanism
Step I Formation of protonated alcohol
Mechanism
Step I Formation of protonated alcohol
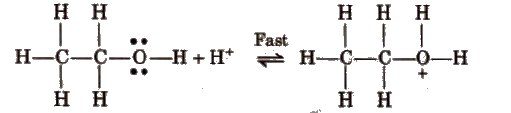 Step II Formation of carbocation
Step II Formation of carbocation
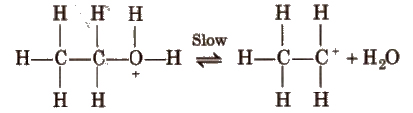 Step III Formation of ethene by elimination of a proton
Step III Formation of ethene by elimination of a proton
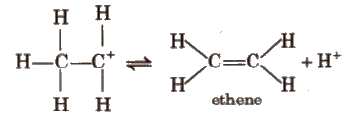 In dehydration reaction, highly substituted alkene is the major product and if the major product is capable of showing cis-trans isomerism, trans-product is the major product. (Saytzeff’s rule).
(iii) Oxidation reactions Oxidising reagents used for the oxidation of alcohols are neutral, acidic or alkaline KMnO4 and acidified K2Cr2O7.
In dehydration reaction, highly substituted alkene is the major product and if the major product is capable of showing cis-trans isomerism, trans-product is the major product. (Saytzeff’s rule).
(iii) Oxidation reactions Oxidising reagents used for the oxidation of alcohols are neutral, acidic or alkaline KMnO4 and acidified K2Cr2O7.
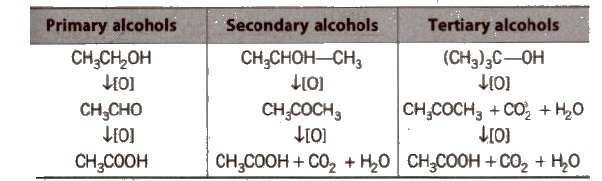 A common reagent that selectively oxidises a primary alcohol to an aldehyde (and no further) is pyridinium chlorochromate (pCC).
A common reagent that selectively oxidises a primary alcohol to an aldehyde (and no further) is pyridinium chlorochromate (pCC).
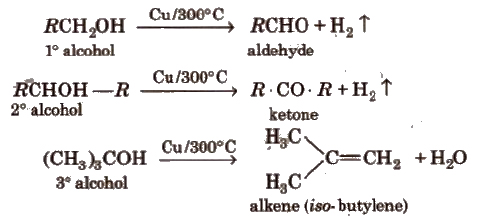
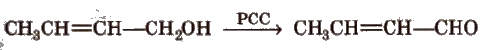 (iv) Dehydrogenation
(iv) Dehydrogenation
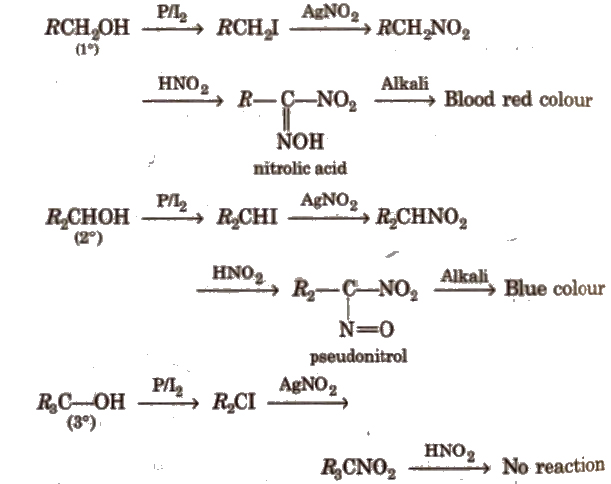
 Distinction among 1°,2° and 3° Alcohols
1°, 2° and 3° alcohols are distinguished by Lucas test, oxidation and reduced copper.
Victor Meyer’s test is also used to distinguish them.
In this test, primary (1°) alcohols give red colour, secondary (2°) alcohols give blue colour and tertiary (3°) alcohols give no colouration.
Distinction among 1°,2° and 3° Alcohols
1°, 2° and 3° alcohols are distinguished by Lucas test, oxidation and reduced copper.
Victor Meyer’s test is also used to distinguish them.
In this test, primary (1°) alcohols give red colour, secondary (2°) alcohols give blue colour and tertiary (3°) alcohols give no colouration.
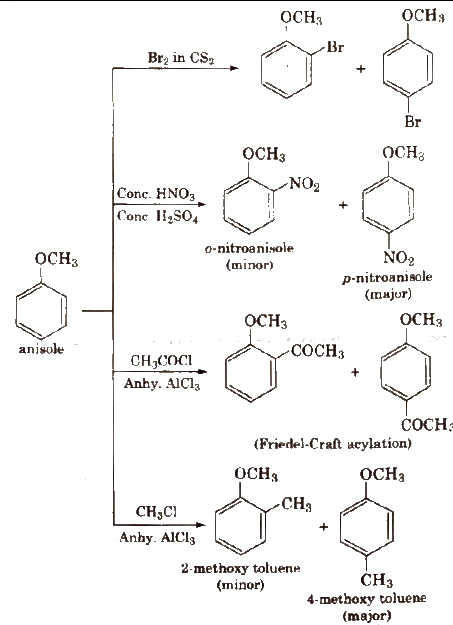
 Reactions of Phenols
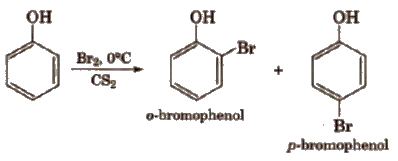
(i) Electrophilic substitution reactions The -OH group attached to the benzene ring activates it towards electrophilic substitution at ortho and para positions .
(a) Halogenation
Reactions of Phenols
(i) Electrophilic substitution reactions The -OH group attached to the benzene ring activates it towards electrophilic substitution at ortho and para positions .
(a) Halogenation
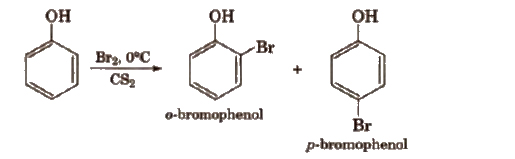
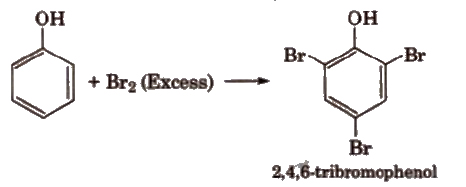 With calculated amount of Br2 in CS2 or CHCI3 it gives ortho and para product.
With calculated amount of Br2 in CS2 or CHCI3 it gives ortho and para product.
 (b) SuLphonation
(b) SuLphonation
 (c) Nitration
(c) Nitration
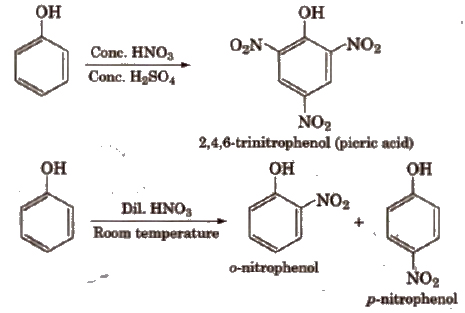 The ortho and para isomers can be separated by steam distillation. This is because o-nitrophenol is steam volatile due to intramolecular hydrogen bonding while p nitrophenol is less volatile due to intermolecular hydrogen bonding which causes the association of molecules.
The ortho and para isomers can be separated by steam distillation. This is because o-nitrophenol is steam volatile due to intramolecular hydrogen bonding while p nitrophenol is less volatile due to intermolecular hydrogen bonding which causes the association of molecules.
 (d) Reimer-Tiemann reaction
(d) Reimer-Tiemann reaction
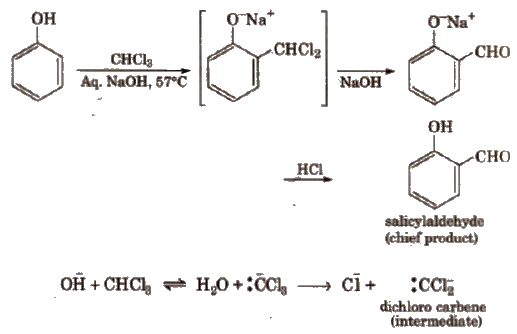 This reaction is an electrophilic substitution reaction and electrophile is dichlorocarbene.
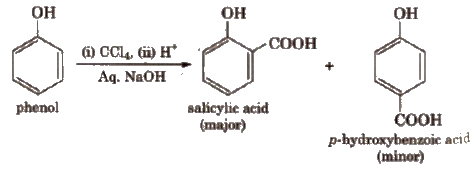
Similarly with carbon tetrachloride and alkali, c- and p-hydroxybenzoic acid are obtained
This reaction is an electrophilic substitution reaction and electrophile is dichlorocarbene.
Similarly with carbon tetrachloride and alkali, c- and p-hydroxybenzoic acid are obtained
 (ii) Kolbe’s reaction
(ii) Kolbe’s reaction
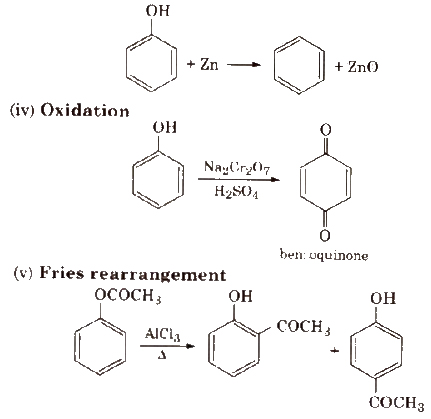
 (iii) Reaction with zinc dust
(iii) Reaction with zinc dust
 Terms Related to Alcohols
(a) Rectified spirit It contains 95% ethyl alcohol and 45% water. It is an azeotrope (constant boiling mixture) and boils at 74°(.
(b) Absolute alcohol Alcohol containing no water, i.e; 100% C2H5OH is known as absolute alcohol. It is prepared as follows.
(i) Quick lime process
(ii) Azeotropic method
(c) Methylated spirit The rectified spirit rendered poisonous by addition of 4-5% methyl alcohol, traces of pyridine and some copper sulphate and is known as methylated spirit or denatured alcohol.
(d) Power alcohol Alcohol mixed with petrol or fuel and used In internal combustion engines Is known as power alcohol.
(e) Wood spirit Methyl alcohol (CH3OH) is also called wood spirit. It is obtained by destructive distillation of wood. Pyroligneous add, the product of destructive distillation of wood, contains acetic acid (10%), methyl alcohol (25%) and acetone (05%). Drinking of methanol causes blindness.
(f) Grain alcohol Ethyl alcohol C2H5OH is also called grain alcohol. It is used In the preparation of various beverages containing different percentages.
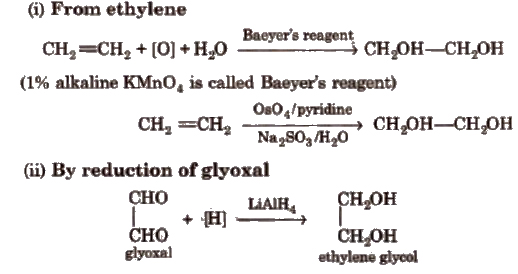
Dihydric Alcohols
These are generally called glycols because of their sweet taste. Ethylene glycol (CH2OH – CH2OH) is the first and most important member of dihydric alcohol series.
Methods of Preparation
Terms Related to Alcohols
(a) Rectified spirit It contains 95% ethyl alcohol and 45% water. It is an azeotrope (constant boiling mixture) and boils at 74°(.
(b) Absolute alcohol Alcohol containing no water, i.e; 100% C2H5OH is known as absolute alcohol. It is prepared as follows.
(i) Quick lime process
(ii) Azeotropic method
(c) Methylated spirit The rectified spirit rendered poisonous by addition of 4-5% methyl alcohol, traces of pyridine and some copper sulphate and is known as methylated spirit or denatured alcohol.
(d) Power alcohol Alcohol mixed with petrol or fuel and used In internal combustion engines Is known as power alcohol.
(e) Wood spirit Methyl alcohol (CH3OH) is also called wood spirit. It is obtained by destructive distillation of wood. Pyroligneous add, the product of destructive distillation of wood, contains acetic acid (10%), methyl alcohol (25%) and acetone (05%). Drinking of methanol causes blindness.
(f) Grain alcohol Ethyl alcohol C2H5OH is also called grain alcohol. It is used In the preparation of various beverages containing different percentages.
Dihydric Alcohols
These are generally called glycols because of their sweet taste. Ethylene glycol (CH2OH – CH2OH) is the first and most important member of dihydric alcohol series.
Methods of Preparation
 Physical Properties
1. It is a colourless, syrupy liquid with sweet taste.
2. Because of its tendency of formation of H-bonds, it is miscible with H2O and ethanol but not with ether.
Chemical Properties
It gives all the general reactions of -OH group.
Physical Properties
1. It is a colourless, syrupy liquid with sweet taste.
2. Because of its tendency of formation of H-bonds, it is miscible with H2O and ethanol but not with ether.
Chemical Properties
It gives all the general reactions of -OH group.
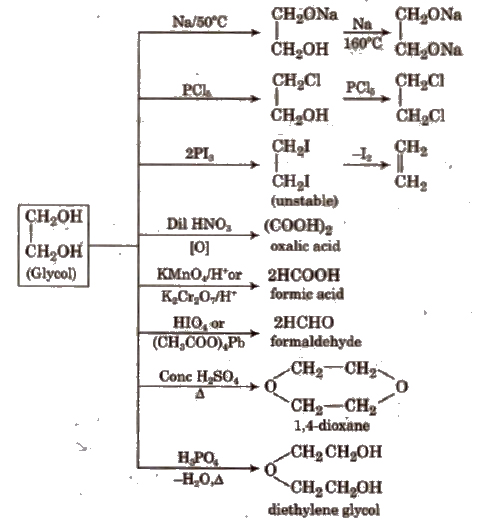 The per-iodic acid cleavage of 1,2-g1ycols is sometimes called Malaprade reaction.
Trihydric Alcohols
Glycerol or glycerine, CH2OH – CH(OH)- CH2OH is the first member of this group. Its IUPAC name is propane-l,2,3-triol.
Method of Preparation
It is obtained as a by product in saponification reaction.
The per-iodic acid cleavage of 1,2-g1ycols is sometimes called Malaprade reaction.
Trihydric Alcohols
Glycerol or glycerine, CH2OH – CH(OH)- CH2OH is the first member of this group. Its IUPAC name is propane-l,2,3-triol.
Method of Preparation
It is obtained as a by product in saponification reaction.
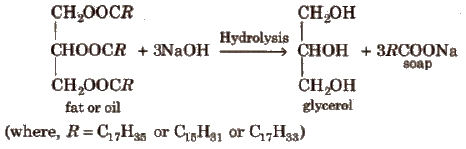 Physical Properties
1. It is a colourless, odourless, viscous and hygroscopic liquid.
2. It is sweet in taste and steam volatile.
3. It is soluble in water but insoluble in ether.
4. Due to excessive H-bonding, it is highly viscous and has high boiling point.
Chemical Properties
It gives all the general reactions given by -OR group but 2° OR is less reactive as compared to 1° .
Some of its specific reactions are :
Physical Properties
1. It is a colourless, odourless, viscous and hygroscopic liquid.
2. It is sweet in taste and steam volatile.
3. It is soluble in water but insoluble in ether.
4. Due to excessive H-bonding, it is highly viscous and has high boiling point.
Chemical Properties
It gives all the general reactions given by -OR group but 2° OR is less reactive as compared to 1° .
Some of its specific reactions are :
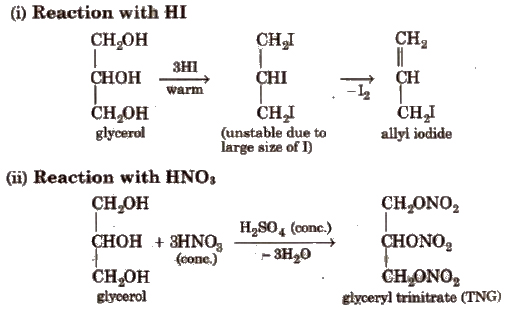
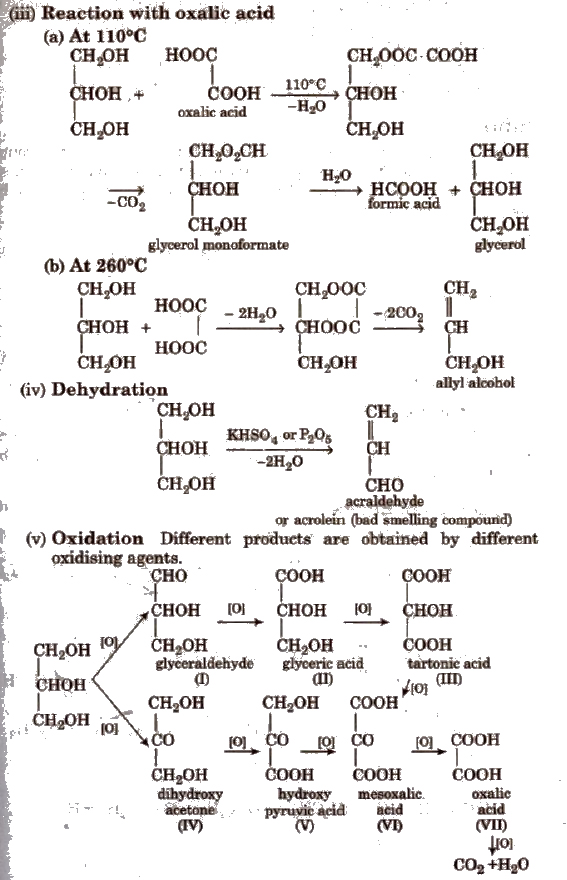 Cone HNO3 gives II; dil HNO3 gives II and III; Bi(NO3)3 or NaNO3 gives VI; Fenton’s reagent or NaOBr or Br2 water in Na2CO3 gives a mixture of I and IV.
Solid KMnO4 oxidises glycerol to VII and CO2 and H2O.
With HIO4 (periodic acid). glycerol gives HCOOH and HCHO.
Ethers
Ethers are the organic compounds in which two alkyl or aryl groups are attached to a divalent oxygen. known as ethereal oxygen. These are represented by the general formula R–O-R” where R may be alkyl or aryl groups. e.g.,
Cone HNO3 gives II; dil HNO3 gives II and III; Bi(NO3)3 or NaNO3 gives VI; Fenton’s reagent or NaOBr or Br2 water in Na2CO3 gives a mixture of I and IV.
Solid KMnO4 oxidises glycerol to VII and CO2 and H2O.
With HIO4 (periodic acid). glycerol gives HCOOH and HCHO.
Ethers
Ethers are the organic compounds in which two alkyl or aryl groups are attached to a divalent oxygen. known as ethereal oxygen. These are represented by the general formula R–O-R” where R may be alkyl or aryl groups. e.g.,
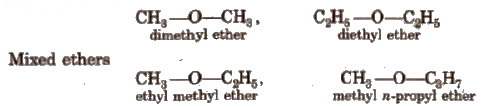 These are the functional isomers of alcohols. These also exhibit chain isomerism and metamerism.
Nomenclature of Ethers
In the IUPAC system, ethers are regarded as ‘alkoxy alkanes’ in which the ethereal oxygen is taken along with smaller alkyl group while the bigger alkyl group is regarded as a part of the alkane.
Preparation of Ethers
These are the functional isomers of alcohols. These also exhibit chain isomerism and metamerism.
Nomenclature of Ethers
In the IUPAC system, ethers are regarded as ‘alkoxy alkanes’ in which the ethereal oxygen is taken along with smaller alkyl group while the bigger alkyl group is regarded as a part of the alkane.
Preparation of Ethers
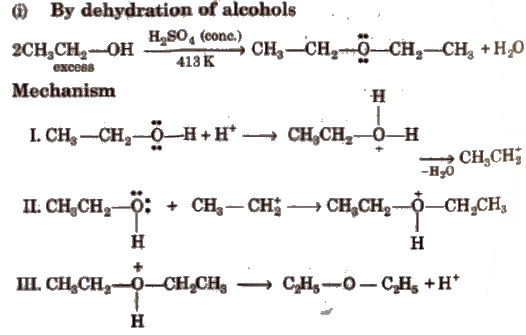 (ii) Williamson’s synthesis Only primary alkyl halides when react with sodium alkoxide give ether while tertiary alkyl halides give alkene due to steric hindrance.
(ii) Williamson’s synthesis Only primary alkyl halides when react with sodium alkoxide give ether while tertiary alkyl halides give alkene due to steric hindrance.
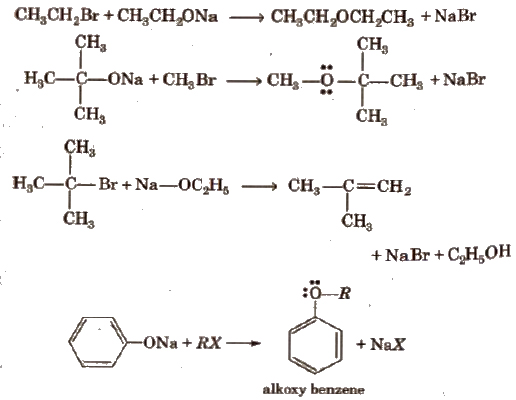 Physical Properties of Ethers
Ethers are polar but insoluble inH20 and have low boiling point than alcohols of comparable molecular masses because ethers do not form hydrogen bonds with water.
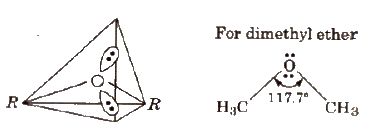
Structure of Ether
The hybridisation of 0 atom in ethers is sp3 (tetrahedral) and its shape is V-shape.
Physical Properties of Ethers
Ethers are polar but insoluble inH20 and have low boiling point than alcohols of comparable molecular masses because ethers do not form hydrogen bonds with water.
Structure of Ether
The hybridisation of 0 atom in ethers is sp3 (tetrahedral) and its shape is V-shape.
 Chemical Reactions of Ether
(i) Reaction with HX
Chemical Reactions of Ether
(i) Reaction with HX
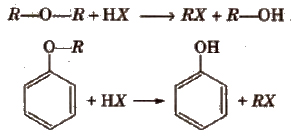 Ethers with two different alkyl groups are also cleaved in the same manner and results in the formation of a primary halide (or smaller and less complex alkyl halide) by SN2 mechanism.
R-O-R’ + HX → RX + R’OR
The order of reactivity of hydrogen halides is as follows
HI > HBr > HCl
In ethers if one of the alkyl groups is a tertiary group, the halide formed is a tertiary halide by SN1 mechanism.
Ethers with two different alkyl groups are also cleaved in the same manner and results in the formation of a primary halide (or smaller and less complex alkyl halide) by SN2 mechanism.
R-O-R’ + HX → RX + R’OR
The order of reactivity of hydrogen halides is as follows
HI > HBr > HCl
In ethers if one of the alkyl groups is a tertiary group, the halide formed is a tertiary halide by SN1 mechanism.
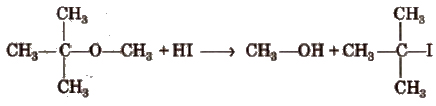 (ii) Halogenation
(ii) Halogenation
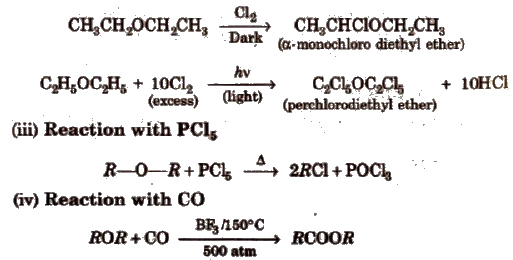 (v) Electrophilic 8ublititutioD reactions In ethers,-OR is ortho, para directing group and activate. the aromatic ring towards electrophilic substitution reaction.
(v) Electrophilic 8ublititutioD reactions In ethers,-OR is ortho, para directing group and activate. the aromatic ring towards electrophilic substitution reaction.
 Ethyl phenyl ester C6H5OC2H5 is also, known as phenetole.
Uses of Ethers
1. Dimethyl ether is used as refrigerant and as a solvent at low temperature.
2. Diethyl Ether is used as an anaesthesia in surgery .
Ethyl phenyl ester C6H5OC2H5 is also, known as phenetole.
Uses of Ethers
1. Dimethyl ether is used as refrigerant and as a solvent at low temperature.
2. Diethyl Ether is used as an anaesthesia in surgery .
Class 12 Key Points, Important Questions & Practice Papers
Hope these notes helped you in your schools exam preparation. Candidates can also check out the Key Points, Important Questions & Practice Papers for various Subjects for Class 12 in both Hindi and English language form the link below.Class 12 NCERT Solutions
Candidates who are studying in Class 12 can also check Class 12 NCERT Solutions from here. This will help the candidates to know the solutions for all subjects covered in Class 12th. Candidates can click on the subject wise link to get the same. Class 12 Chapter-wise, detailed solutions to the questions of the NCERT textbooks are provided with the objective of helping students compare their answers with the sample answers.Class 12 Mock Test / Practice
Mock test are the practice test or you can say the blue print of the main exam. Before appearing in the main examination, candidates must try mock test as it helps the students learn from their mistakes. With the help of Class 12 Mock Test / Practice, candidates can also get an idea about the pattern and marking scheme of that examination. For the sake of the candidates we are providing Class 12 Mock Test / Practice links below.Class 12 Exemplar Questions
Exemplar Questions Class 12 is a very important resource for students preparing for the Examination. Here we have provided Exemplar Problems Solutions along with NCERT Exemplar Problems Class 12. Question from very important topics is covered by Exemplar Questions for Class 12. Class 12 Chemistry Maths Notes Physics Notes Biology NotesTo get study material, exam alerts and news, join our Whatsapp Channel.




